FELIX papers of the Ryzhov group
Papers of the Ryzhov
group (NIU) showing the results of several FELIX measurement campaigns:
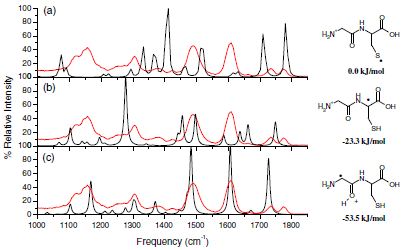
 Structure and Reactivity of the N-Acetyl-Cysteine Radical Cation and Anion: Does Radical Migration Occur?
Structure and Reactivity of the N-Acetyl-Cysteine Radical Cation and Anion: Does Radical Migration Occur?
Sandra Osburn, Giel Berden, Jos Oomens, Richard A. J. O’Hair, and Victor Ryzhov
The structure and reactivity of the N-acetyl-cysteine radical cation and anion were studied using ion-molecule reactions, infrared multi-photon dissociation (IRMPD) spectroscopy, and density functional theory (DFT) calculations. The radical cation was generated by first nitrosylating the thiol of N-acetyl-cysteine followed by the homolytic cleavage of the S–NO bond in the gas phase. IRMPD spectroscopy coupled with DFT calculations revealed that for the radical cation the radical migrates from its initial position on the sulfur atom to the a-carbon position, which is 2.5 kJ mol–1 lower in energy. The radical migration was confirmed by time-resolved ion-molecule reactions. These results are in contrast with our previous study on cysteine methyl ester radical cation and the study by Sinha et al. for cysteine radical cation where the radical was found to stay on the sulfur atom as formed. A similar approach allowed us to form a hydrogen-deficient radical anion of N-acetyl-cysteine, (M – 2H) •– . IRMPD studies and ion-molecule reactions performed on the radical anion showed that the radical remains on the sulfur, which is the initial and more stable (by 63.6 kJ mol–1) position, and does not rearrange. © 2011 American Society for Mass Spectrometry.
PDF file: download here
Reference: Journal of The American Society for Mass Spectrometry 22 (2011) 1794-1803.
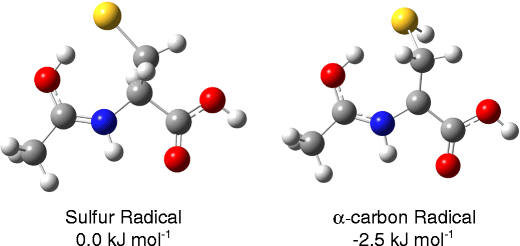
 S-to-alphaC Radical Migration in the Radical Cations of Gly-Cys and Cys-Gly.
S-to-alphaC Radical Migration in the Radical Cations of Gly-Cys and Cys-Gly.
Sandra Osburn, Giel Berden, Jos Oomens, Richard A. J. O’Hair, and Victor Ryzhov
The radical cations of Cys-Gly and Gly-Cys were studied using ion-molecule reactions (IMR), infrared multiple-photon dissociation (IRMPD) spectroscopy, and density functional theory (DFT) calculations. Homolytic cleavage of the S–NO bond of nitrosylated precursors generated radical cations with the radical site initially located on the sulfur atom. Time-resolved ion-molecule reactions showed that radical site migration via hydrogen atom transfer (HAT) occurred much more quickly in Gly-Cys•+ than in Cys-Gly•+. IRMPD and DFT calculations indicated that for Gly-Cys, the radical migrated from the sulfur atom to the a-carbon of glycine, which is lower in energy than the sulfur radical (–53.5 kJ/mol). This migration does not occur for Cys-Gly because the glycine a-carbon is higher in energy than the sulfur radical (10.3 kJ/mol). DFT calculations showed that the highest energy barriers for rearrangement are 68.2 kJ/mol for Gly-Cys and 133.8 kJ/mol for Cys-Gly, which is in agreement with both the IMR and IRMPD data and explains the HAT in Gly-Cys.© 2012 American Society for Mass Spectrometry.
PDF file: download here
Reference: Journal of The American Society for Mass Spectrometry 23 (2012) 1019-1023.
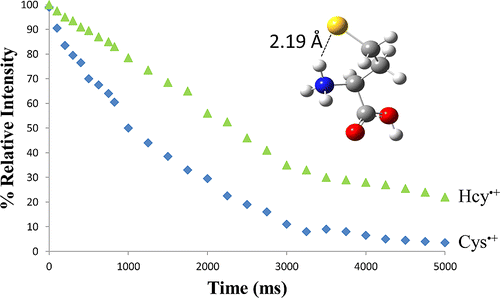 Structure and Reactivity of Homocysteine Radical Cation in the Gas Phase Studied by Ion–Molecule Reactions and Infrared Multiple Photon Dissociation.
Structure and Reactivity of Homocysteine Radical Cation in the Gas Phase Studied by Ion–Molecule Reactions and Infrared Multiple Photon Dissociation.
S. Osburn, T. Burgie, G. Berden, J. Oomens, R. A. J. O’Hair and V. Ryzhov
The reactivity of the cysteine (Cys) and homocysteine (Hcy) radical cation was studied using ion–molecule reactions. The radical cations were generated via collision-induced dissociation (CID) of their S-nitrosylated precursors. Cleavage of the S–NO bond led to the formation of the radical initially positioned on the sulfur atom. The reactions of the radical cations with dimethyl disulfide revealed that the cysteine radical cation reacts more quickly than the homocysteine radical cation. Infrared multiple photon dissociation (IRMPD) spectroscopy and density functional theory (DFT) calculations were used to determine the structure of the homocysteine radical cation, which was compared to the previously published structure of the cysteine radical cation (Sinha et al. Phys. Chem. Chem. Phys.2010, 12, 9794–9800). IRMPD spectroscopy and DFT calculations revealed that this difference in radical reactivity was not a result of a radical rearrangement for the homocysteine radical cation but rather that the reactivity was modulated by stronger hydrogen bonding. © 2013 American Chemical Society.
PDF file: download here
Reference: Journal of Physical Chemistry A 117 (2013) 1144-1150
Identification of novel fragmentation pathways and fragment ion structures in the tandem mass spectra of protonated synthetic cathinones
See Forensic Chemistry (2020): https://doi.org/10.1016/j.forc.2020.100245
Last update: 2016
 Structure and Reactivity of the N-Acetyl-Cysteine Radical Cation and Anion: Does Radical Migration Occur?
Structure and Reactivity of the N-Acetyl-Cysteine Radical Cation and Anion: Does Radical Migration Occur? S-to-alphaC Radical Migration in the Radical Cations of Gly-Cys and Cys-Gly.
S-to-alphaC Radical Migration in the Radical Cations of Gly-Cys and Cys-Gly. Structure and Reactivity of Homocysteine Radical Cation in the Gas Phase Studied by Ion–Molecule Reactions and Infrared Multiple Photon Dissociation.
Structure and Reactivity of Homocysteine Radical Cation in the Gas Phase Studied by Ion–Molecule Reactions and Infrared Multiple Photon Dissociation.